 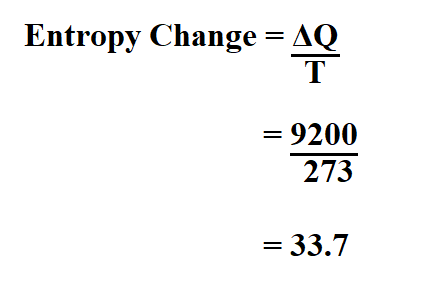
We write \(S^o_A\left(T\right)\) to indicate the absolute entropy of substance \(A\) in its standard state at temperature \(T\). where p is the pressure and V is the volume of the gas. Entropy change what you end up with - what you started with. Substituting for the definition of work for a gas. You ended up with 1 mole of carbon dioxide and two moles of liquid water. Using standard absolute entropies at 298K, calculate. where E is the internal energy and W is the work done by the system. Learn more about entropy and understand how to use the entropy equation through the example calculation. We begin by using the first law of thermodynamics: dE dQ - dW. It is usually included in compilations of thermodynamic data for chemical substances. For gases, there are two possible ways to evaluate the change in entropy. Explanation QUESTION QUESTION An oxygen O2. Calculate the entropy of a string by determining the frequency of each character, then summing each characters probability multiplied by the log base 2 of that. The few simple steps required for the calculation of the absolute entropy are (1) Run CREST in default mode on a starting structure to find the lowest conformer. 
The standard entropy is usually given the symbol \(S^o\). View Calculating absolute entropy using the Boltzmann hypothesis.pdf from CHEM 1212K at Georgia Gwinnett College. When the entropy value is calculated for one mole of the substance in its standard state, the resulting absolute entropy is called the standard entropy. Only a perfectly ordered, crystalline substance at absolute zero would exhibit no. Vibrational, rotational, and translational motions of a carbon dioxide molecule are illustrated here. Where the substance undergoes phase changes, the contribution that the phase change makes to the entropy of the substance is equal to the enthalpy change for the phase change divided by the temperature at which it occurs.Īt any given temperature, the entropy value that is obtained in this way is called the substance’s absolute entropy or its third-law entropy. The entropy of any perfectly ordered, crystalline substance at absolute zero is zero. 
Phase changes are isothermal and reversible. In temperature ranges where experimental heat capacity data are available, the entropy change is obtained by integration using these data. \), using Debye’s theoretical relationship, \(C_P=AT^3\) \(A\) is obtained from the value of \(C_P\) at the lowest temperature for which an experimental value of \(C_P\) is available. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
